Cores
- Transgenic and CRISPR Mouse Core
- Metabolic and Molecular Physiology Core
- Genomics & Epigenetics Core
- Human Genetics Core
- Biological Imaging Core
Core Director: Pamela L. Mellon, PhD
Professor of Obstetrics, Gynecoology, and Reproductive Sciences and Neurosciences
Core Co-Director: Miles F. Wilkinson, PhD
Professor of Obstetrics, Gynecoology, and Reproductive Sciences
 The DRC Transgenic, CRISPR Mutagenesis, and Knock-out Mouse Core is a state-of-the-art facility that has an outstanding track record in the production of genetically altered mice. Transgenic mice carrying new or novel genes are created by microinjection of DNA into the pronuclei of fertilized eggs.Mice carrying directed mutations are created by injection of CRISPR guide RNAs with Cas9 mRNA/protein and template DNA. Knock-out mice lacking specific genes of interest are created by homologous recombination in embryonic stem cells followed by injection into blastocysts to create chimeric mice. Highly experienced personnel produce transgenic, CRISPR, and knock-out mice for DRC investigators at very reasonable cost and with very short lead times. The Core provides embryonic stem cell recombination, knockout mice, CRISPR mutant mice, transgenic mice (both standard and BAC transgenics), sperm or embryo cryopreservation, and pathogen-free embryonic rederivation to the DRC community at discounted rates. This UCSD-based Core Facility has been in operation since 1992.
The DRC Transgenic, CRISPR Mutagenesis, and Knock-out Mouse Core is a state-of-the-art facility that has an outstanding track record in the production of genetically altered mice. Transgenic mice carrying new or novel genes are created by microinjection of DNA into the pronuclei of fertilized eggs.Mice carrying directed mutations are created by injection of CRISPR guide RNAs with Cas9 mRNA/protein and template DNA. Knock-out mice lacking specific genes of interest are created by homologous recombination in embryonic stem cells followed by injection into blastocysts to create chimeric mice. Highly experienced personnel produce transgenic, CRISPR, and knock-out mice for DRC investigators at very reasonable cost and with very short lead times. The Core provides embryonic stem cell recombination, knockout mice, CRISPR mutant mice, transgenic mice (both standard and BAC transgenics), sperm or embryo cryopreservation, and pathogen-free embryonic rederivation to the DRC community at discounted rates. This UCSD-based Core Facility has been in operation since 1992.
Transgenic and Knockout Mouse Core:
Moores Cancer Center Transgenic MouseThis form must be filled out before beginning any work with the Core: Transgenic Core Request For Services

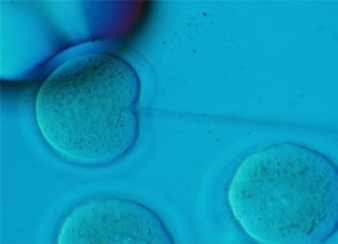 A desired DNA construct is injected into the pronucleus of a 0.5 d.p.c. embryo. Resulting pups are returned to the client. The client will then analyze these by PCR or Southern blot to determine the presence of the given transgene.
A desired DNA construct is injected into the pronucleus of a 0.5 d.p.c. embryo. Resulting pups are returned to the client. The client will then analyze these by PCR or Southern blot to determine the presence of the given transgene.
For internal deletion, a CRISPR guide RNA that targets the desired gene region is coinjected with commercial Cas9 mRNA or protein. For specific mutations or insertion/deletions, a CRISPR guide RNA that targets the desired gene region is coinjected with commercial Cas9 mRNA or protein and a template DNA that harbors the mutation. Generally, these are injected into the cytoplasm of fertilized one-cell embryos. Resulting pups are returned to the client. The client will then analyze these by PCR and sequencing to determine the presence of the given transgene.
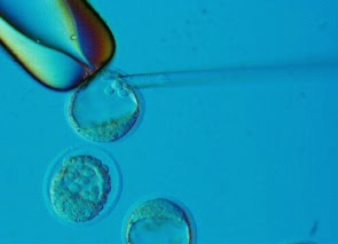 Mouse embryonic stem cells that have undergone homologous recombination are injected into the blastocoel cavity of 3.5 d.p.c. blastocyst stage C57Bl6 (black or albino as needed) embryos. Injected blastocysts are implanted into pseudopregnant recipient females, and chimeric pups are born. Chimerics are returned to the client. Those that transmit the 'knock-out' allele to germline produce agouti pups, which are analyzed for phenotype by the client.
Mouse embryonic stem cells that have undergone homologous recombination are injected into the blastocoel cavity of 3.5 d.p.c. blastocyst stage C57Bl6 (black or albino as needed) embryos. Injected blastocysts are implanted into pseudopregnant recipient females, and chimeric pups are born. Chimerics are returned to the client. Those that transmit the 'knock-out' allele to germline produce agouti pups, which are analyzed for phenotype by the client.
A targeting vector containing a 'knock-out' or 'knock-in' gene of interest is electroporated into mouse embryonic stem cells (C57Bl6). The cells undergo drug selection, clones are picked and expanded in tissue culture, then returned to the client for analysis. Homologous Recombinant clones are verified by the client using either PCR or Southern Blot analysis by the client.

Chromosome counting is done on targeted homologous recombinant ES cell clones prior to blastocyst injection for quality control.
Embryo rederivation is used to create a pathogen-free strain of mice from lines that arrive at the University from a non-approved vendor and mice contaminated with various style="margin-top: 5px; margin-bottom: 15px; margin-left: 20px;"pathogens (MHV, Pinworm, Parvovirus). Males arrive in the Rodent Isolation Unit (RIU), and wild-type females of the desired background strain are superovulated and mated to the males. Embryos are harvested in the RIU, washed and transferred into pathogen free, pseudopregnant recipient females, which are returned to the client.
In vitro fertilization (IVF) can be used to resuscitate a mouse line from cryopreserved sperm, jump start a breeding colony, or to perform characterization studies on strains with poor breeding efficiency. We need one healthy live male or we are able to resuscitate the line using cryopreserved sperm from our facility or cryopreserved sperm shipped in from other sourceds. Success rate depends on the quality of sperm provided and the background strain.
Genetic banking through the use of mouse embryo and sperm cryopreservation is a practical means to store scientifically valuable mice. Mouse embryos or sperm stored in liquid nitrogen offer a safe way to preserve lines with potential future use but no current use and offer savings in facility space and the expense associated with keeping a breeding colony. Another advantage is that during the process of embryo or sperm collection, most pathogenic organisms are excluded as in other methods used to rederive lines of mice.
Briefly, the embryo cryopreservation procedure entails harvesting embryos from donor females at the 8-cell developmental stage that is optimal for cryopreservation by our method. A controlled rate methanol bath freezer is used to gradually cool the embryos to -80°C, before plunging them into liquid nitrogen for storage. Quality control is assured by freezing and thawing a representative sample of wild-type frozen embryos along with each line frozen down. Frozen embryos should be retrieved by the investigator one week post-freezing for long-term storage in liquid nitrogen in the investigator's own lab.
Alternatively, sperm can be cryopreserved. In this case, at least two proven male breeders between the ages of 3-5 months are provided to the Core. Core cryopreserves sperm and stores in LN2 for the client. Client does long-term storage in LN2.

Our CRISPR/Cas9 consultation service includes guide RNA design, donor DNA selection and design, and Cas9 selection. The gRNA design serivce also includes gRNA design of CRISPR/Cas9, CRISPR/Cas12a, and prime editing using pegRNAs. Additionally, our consultation service provides primer design for genotyping, review and torubleshooting of the genotyping results, and suggests a breeding plan. If clients are not familiar with CRISPR/Cas9, the consultation serices providews the guidelines and methods for preparation of the mixture for mircroinjection.
The DRC rates are the same as those shown for Cancer Center Members. See our detailed website for our current rates: Moores Cancer Center Transgenic Mouse
Sang Lee, PhD
srl001@health.ucsd.edu
Ryan Fiske
rfiske@health.ucsd.edu